- Sample stability (30 days at room temperature)
- Sample neutrality (unbiased representation of the in vivo state)
- Reproducibility (lot-to-lot and aliquot-to-aliquot)
- Easy to use for improved donor compliance
- Compatibility with all sample types (adult and pediatric cohorts)

With over 20 years of trusted experience, DNA Genotek, a subsidiary of OraSure Technologies, Inc., is a leading provider of non-invasive biological sample collection technology. We are pleased to present the OMNIgene®•GUT Dx kit—the first and only FDA authorized collection device (via De Novo pathway) validated for fecal selfcollection and stabilization of bacterial DNA for gut microbiome profiling.
OMNIgene®•GUT Dx OMD-200
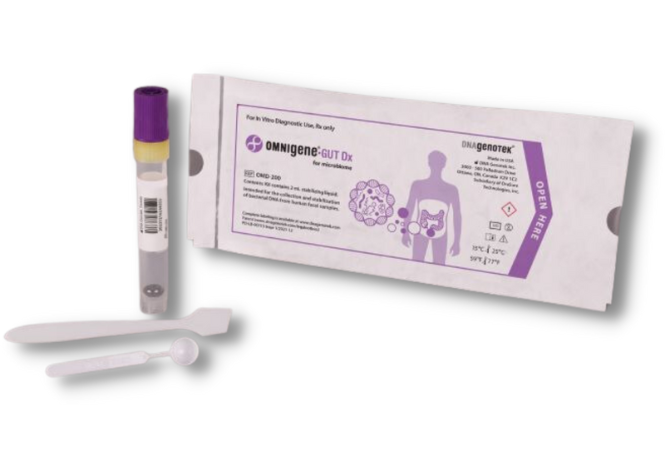
Standardize workflow
Simplify research
Accelerate time to market
Bring confidence and accuracy to your
microbiome discovery
The FDA granted the OMNIgene®•GUT Dx device a De Novo authorization based on a robust validation of:

Reduce validation time, experimental bias and failed collections by leveraging the only validated and FDA authorized device for in vitro diagnostic use.




Inside DNA Genotek
Legal
